|
Which one you choose depends on the data you've collected beforehand. The above formula is one of our calculator's four partial pressure formulas. 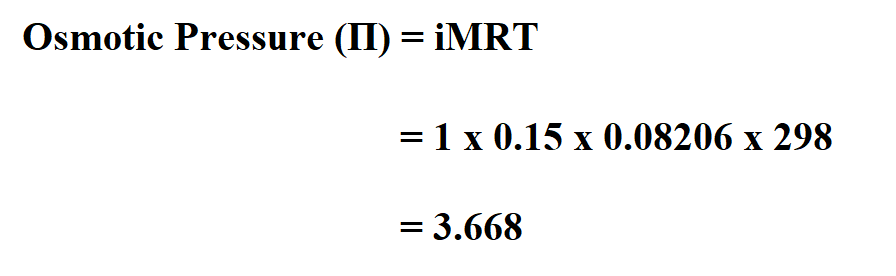
It shows that the partial pressure of one component is proportional to its mole fraction. Where mole fraction is the ratio of moles of the selected gas to the moles of the entire gas mixture. Partial pressure = total pressure × mole fraction Where p 1, p 2, and so on, up to p n, represent the partial pressure of each gaseous component. It can also be illustrated with an equation: The total pressure exerted on a container's walls by a gas mixture is equal to the sum of the partial pressures of each separate gas. All these chemistry calculators are free to use. We have covered most concepts ranging from acids to bases, Gay Lussac’s law, Boyles law, normality, density, and many other chemistry concepts. The partial pressure of one component of this mixture is the pressure that this individual gas exerts. You can use these online free chemistry calculators as and when you need a helping hand with your chemistry calculations. If a mixture of ideal gases (i.e., where the molecules don't interact with each other) is sealed within a container, the gases will diffuse and fill up all of the available space. The Gas constant is a physical constant that is introduced in many fundamental equations in the physical sciences, for instance the ideal gas law and the Nernst equation… The gas constant also well-known as the molar, universal, or ideal gas constant, represented by the symbol R. Use ideal gas law calculator on the top check the bellow results. Substitute the specified values in the equation: Examples on Ideal Gas Law Calculator with StepsĮxample 1: Ideal gas law solve for moles, pv=nrt solve for n:įind the number of moles at 300K temperature, 250kPa pressure and 5 liters volume? State the equation you plan to use and plug in the values.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
